
Comparison of Ti, Zr, and Hf as Cations for Metallocene-Catalyzed Olefin Polymerization | Organometallics

Zr and Hf have Equal Atomic Radius | lanthanide Contraction | Inorganic #chemistrylearning - YouTube
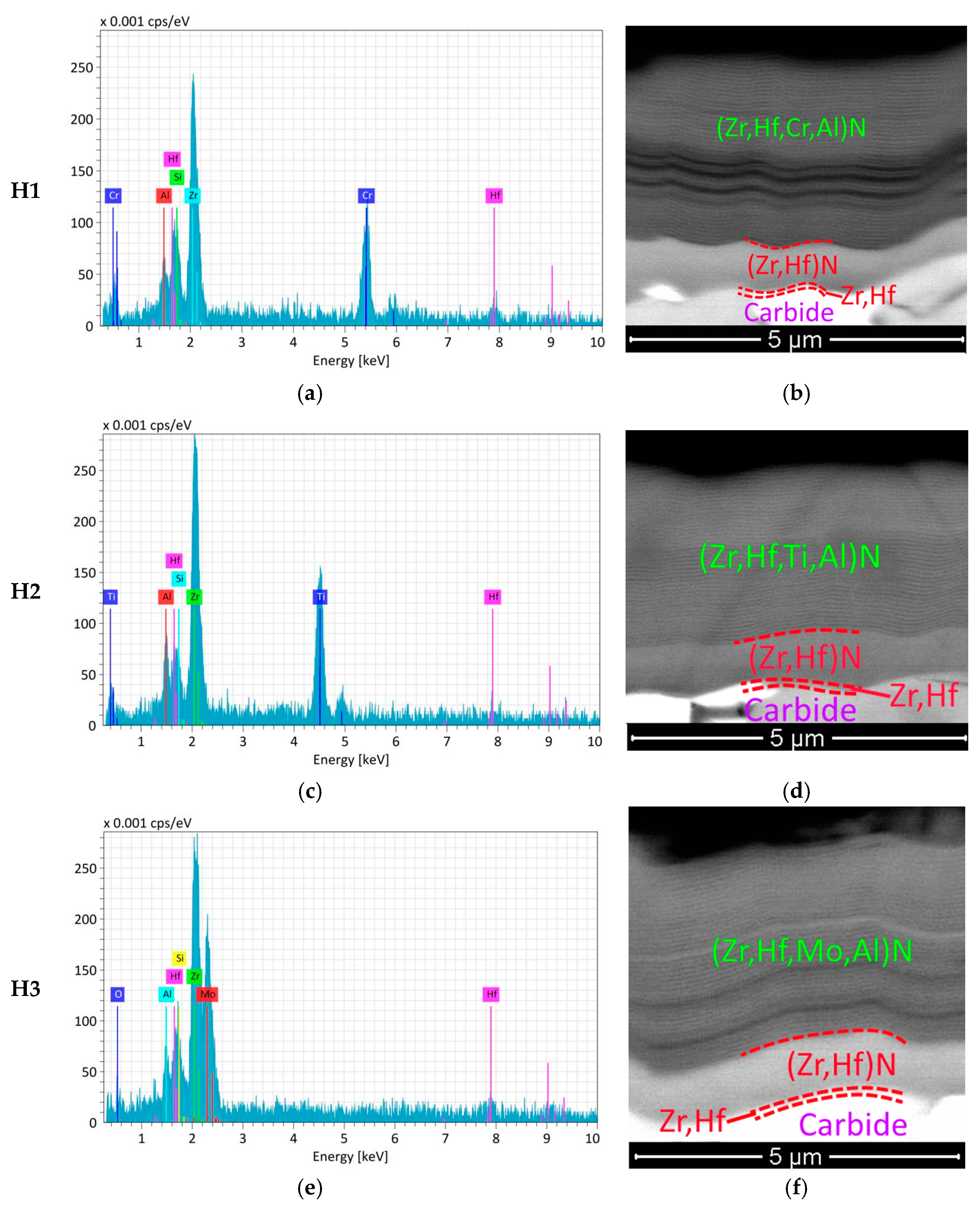
Coatings | Free Full-Text | Investigation of Properties of the Zr,Hf-(Zr,Hf )N-(Zr,Hf,Me,Al)N Coatings, Where Me Means Cr, Ti, or Mo

A): Zr and Hf have nearly equal atomic radii. (R): Zr and Hf belong to the same group. | 12 | D... - YouTube

Due to lanthanide contraction Fe, Co, Ni have equal size Zr and Hf have equal size C All f-block ions have equal size D Zn and Zn+2 have almost equal size

Why `Zr` and `Hf` or `Nb` and `Ta` exhibit similar properties? Or Zirconium (atomic number40) - YouTube

Zr and Hf have Equal Atomic Radius | lanthanide Contraction | Inorganic #chemistrylearning - YouTube





![Kannada] Zr and Hf have almost identical atomic radii. Give reason? Kannada] Zr and Hf have almost identical atomic radii. Give reason?](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/6454334.webp)



![Bengali] Compare atomic radii of Zr (Z = 40) and Hf (Z = 72). Bengali] Compare atomic radii of Zr (Z = 40) and Hf (Z = 72).](https://static.doubtnut.com/ss/web-overlay-thumb/3591037.webp)


![Kannada] Zr and Hf have almost identical atomic radii. Give reason. Kannada] Zr and Hf have almost identical atomic radii. Give reason.](https://static.doubtnut.com/ss/web-overlay-thumb/5540907.webp)





