If there's an air bubble 10km underwater, would it be possible to swim in it due to the compression? - Quora
If there's an air bubble 10km underwater, would it be possible to swim in it due to the compression? - Quora

An air bubble of radius `r` in water is at a depth `h` below the water surface at some instant. If - YouTube
Relationship of underwater depth, gas volume, and gas pressure. Adapted... | Download Scientific Diagram
Is it possible for a bubble of air to be so deep under water that it cannot rise up due to too high pressure? - Quora

The history of an air bubble vibration in water, for frequency 26.5... | Download Scientific Diagram
Is it possible for a bubble of air to be so deep under water that it cannot rise up due to too high pressure? - Quora

Physiology of Deep-Sea Diving and Other Hyperbaric Conditions - Aviation, Space, and Deep-Sea Diving Physiology - Guyton and Hall Textbook of Medical Physiology, 12th Ed

The volume of an air bubble is doubled as it rises from the bottom of a lake to its surface. If the atmospheric pressure is H m of mercury & the density
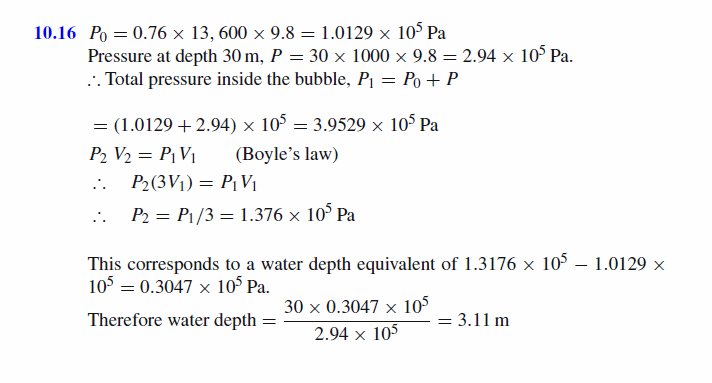
A bubble of gas rises from the bottom of a lake 30 m deep. At what depth will the volume be thrice as great as it was originally (atmospheric pressure = 0.76

A bubble of gas released at the bottom of a lake increases to eight times its original volume when it reaches the surface. Assuming that atmospheric pressure is equivalent to the pressure

The history of an air bubble vibration in water, for frequency 26.5... | Download Scientific Diagram