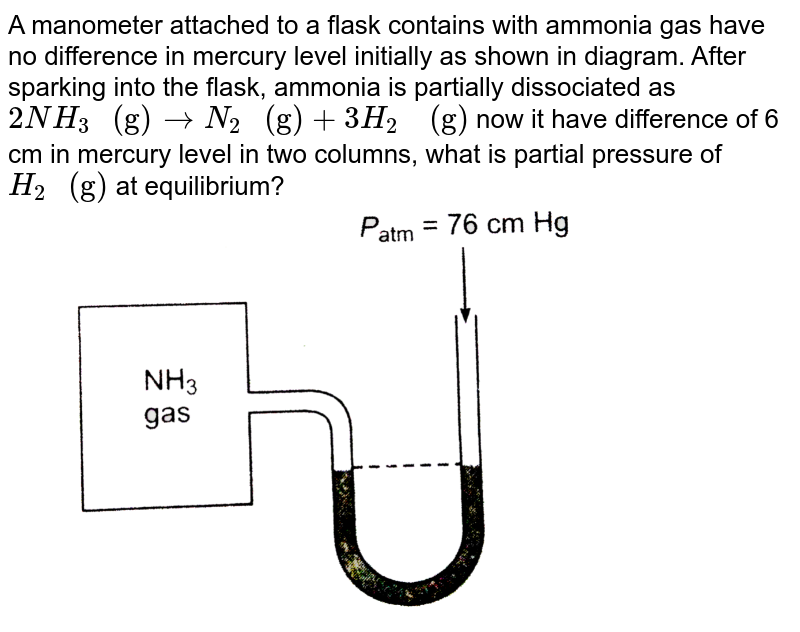
A manometer attached to a flask contains with ammonia gas have no difference in mercury level in... - YouTube

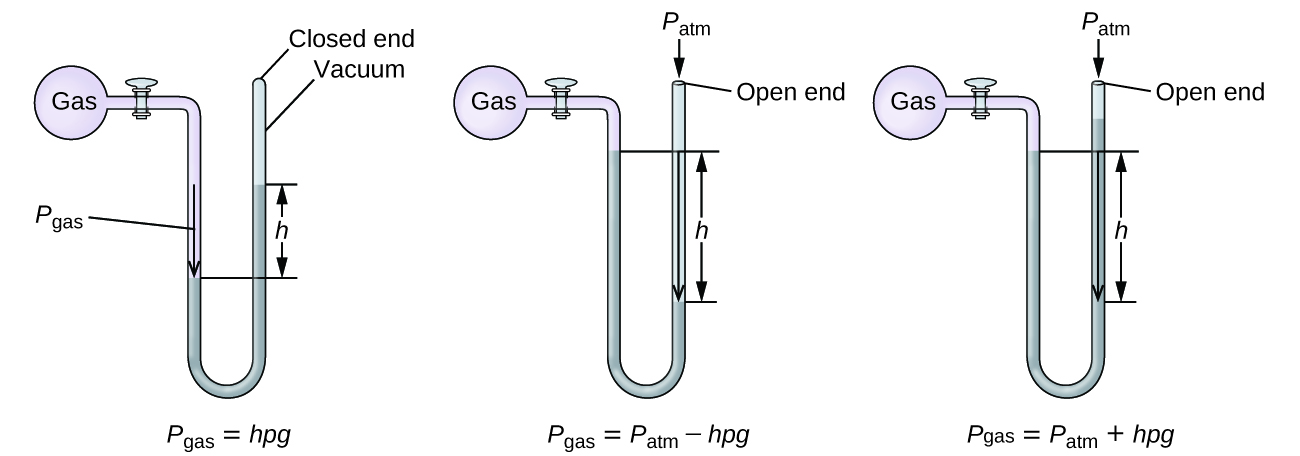
The diagram below shows an open-tube manometer. When the flask is open to the atmosphere, the levels of mercury are equal. For each of the following scenarios, where gas is present in

A manometer attached to a flask contains with ammonia gas have no difference in mercury level in... - YouTube
![a diagram for an open-tube manometer is shown below. [{Image src='diag8594138664026351785.jpg' alt='reaction' caption=''}] | Homework.Study.com a diagram for an open-tube manometer is shown below. [{Image src='diag8594138664026351785.jpg' alt='reaction' caption=''}] | Homework.Study.com](https://homework.study.com/cimages/multimages/16/diag8594138664026351785.jpg)
a diagram for an open-tube manometer is shown below. [{Image src='diag8594138664026351785.jpg' alt='reaction' caption=''}] | Homework.Study.com

Premium Vector | Manometer with gas and liquid in the valve open air and gas pressure test closed and open end manome
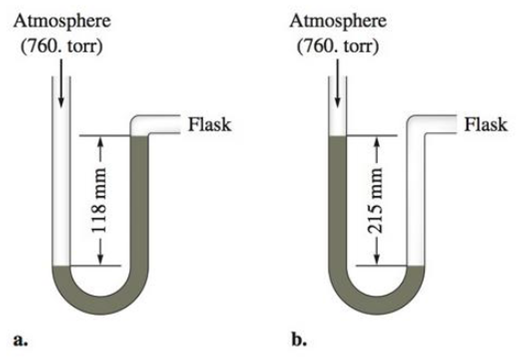
A diagram for an open-tube manometer is shown below. If the flask is open to the atmosphere, the mercury levels are equal. For each of the following situations where a gas is

Manometer Pressure Problems, Introduction to Barometers - Measuring Gas & Atmospheric Pressure - YouTube

Chapter 10 “Gases” A Gas 4 Uniformly fills any container. 4 Mixes completely with any other gas 4 Exerts pressure on its surroundings. 4 (show demo with. - ppt download
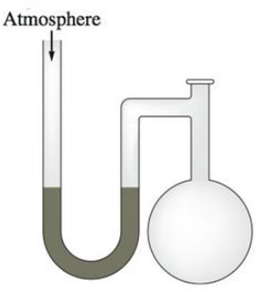
A diagram for an open-tube manometer is shown below. If the flask is open to the atmosphere, the mercury levels are equal. For each of the following situations where a gas is

A sealed-tube manometer as below-mentioned can be used to measure pressures below atmospheric pressure. The tube above the mercury is evacuated. When there is a vacuum in the flask, the mercury levels
A manometer contains a liquid of density 5.44 g/cm is attached to a flask containing gas 'A as follows
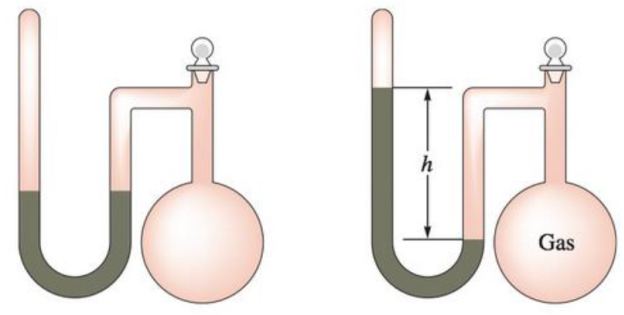
A sealed-tube manometer (as shown below) can be used to measure pressures below atmospheric pressure. The tube above the mercury is evacuated. When there is a vacuum in the flask, the mercury

Gas absorption release reaction flask. a. 20% of potassium hydroxide... | Download Scientific Diagram